scTransform#
As a single-cell RNA sequencing transform method, scTransform uses regularized negative binomial regression to normalize the express matrix of UMI [Hafemeister19].
Differences between methods#
Before exploring scTransform, let’s review what classic normalization does.
[1]:
import sys
import stereo as st
import pandas as pd
import numpy as np
# read data
data1 = st.io.read_gef('./SS200000135TL_D1.tissue.gef')
data1.sparse2array()
gmean = np.exp(np.log(data1.exp_matrix.T + 1).mean(1)) - 1
# preprocessing
data1.tl.raw_checkpoint()
data1.tl.normalize_total(target_sum=1e4)
data1.tl.log1p()
log_normalize_result = pd.DataFrame([gmean, data1.exp_matrix.T.var(1)], index=['gmean', 'log_normalize_variance'], columns=data1.gene_names).T
from stereo.algorithm.sctransform.plotting import plot_log_normalize_var
fig1=plot_log_normalize_var(log_normalize_result)

After log1p normalization, it is apparently observed that lowly expressed genes contribute just a little variance in this sample.
[2]:
data2 = st.io.read_gef('./SS200000135TL_D1.tissue.gef')
data2.tl.sctransform(res_key='sctransform', inplace=True, filter_hvgs=True)
from stereo.algorithm.sctransform.plotting import plot_residual_var
fig2=plot_residual_var(data2.tl.result['sctransform'])
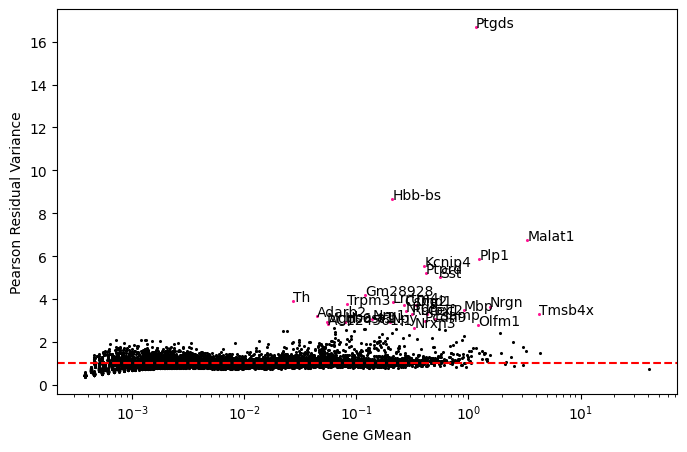
Whereas, after scTransform, gene express matrix is transformed from raw counts to Pearson residual. Different with 1og1p normalization, scTransform balances variance distribution of all genes, which means that not only highly expressed genes make sense, so do the lowly expressed genes.
Let us take some genes from a real dataset after normalization via scTransform, and compare their variance distribution to that normalized by log1p.
[3]:
data3 = st.io.read_gef('./SS200000135TL_D1.tissue.gef')
data3.tl.cal_qc()
data3.plt.spatial_scatter_by_gene(gene_name='Th')
from stereo.algorithm.sctransform.plotting import plot_genes_var_contribution
fig3=plot_genes_var_contribution(data3, gene_names=['Ptgds','Hbb-bs', 'Kcnip4', 'Gm28928', 'Trpm3', 'Th'])
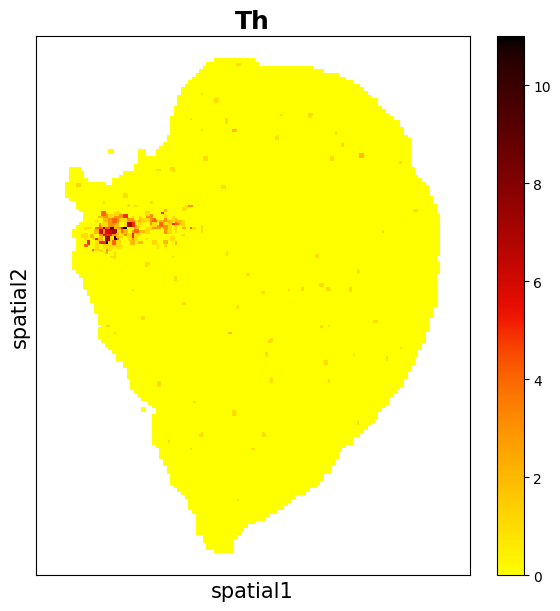
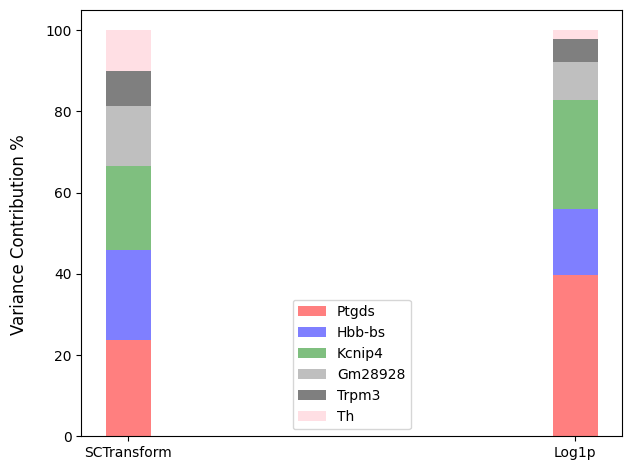
The expression of gene Th is low. In log1p normalization, although variance contribution is reduced, the spots of gene Th are concentrated in a certain region. When using standardization methond of scTransform, the variance contribution can be improved.
Note
More contribution from lowly expressed genes might help us focus on tiny differentiation between subtypes within cell clusters, which is barely found because of over shrinking of log sometimes.
Clustering after different normalizations#
Normalization via scTransform#
For further exploration, let’s try clustering method on these result. Note that scTransform defaults to sampling with 5000 cells and 2000 genes for estimating parameters. If want to fit with more cells, adjust the n_cells and n_genes parameters.
[4]:
import sys
import stereo as st
import time
# read data
data_sct = st.io.read_gef('./SS200000135TL_D1.tissue.gef')
# preprocessing
data_sct.tl.cal_qc()
data_sct.tl.sctransform(res_key='sctransform', inplace=True, filter_hvgs=True, n_cells=5000, n_genes=2000)
# embedding
data_sct.tl.pca(use_highly_genes=False, hvg_res_key='highly_variable_genes', n_pcs=20, res_key='pca', svd_solver='arpack')
data_sct.tl.neighbors(pca_res_key='pca', n_pcs=30, res_key='neighbors', n_jobs=8)
data_sct.tl.umap(pca_res_key='pca', neighbors_res_key='neighbors', res_key='umap', init_pos='spectral', spread=2.0)
# clustering
data_sct.tl.leiden(neighbors_res_key='neighbors', res_key='leiden')
completed 0 / 500 epochs
completed 50 / 500 epochs
completed 100 / 500 epochs
completed 150 / 500 epochs
completed 200 / 500 epochs
completed 250 / 500 epochs
completed 300 / 500 epochs
completed 350 / 500 epochs
completed 400 / 500 epochs
completed 450 / 500 epochs
Get the list of top 10 feature genes.
[5]:
sct_high_genes = data_sct.tl.result['sctransform'][1]['top_features']
sct_high_genes.tolist()[:10]
[5]:
['Ptgds',
'Hbb-bs',
'Malat1',
'Plp1',
'Kcnip4',
'Ptprd',
'Sst',
'Gm28928',
'Th',
'Trpm3']
Normalization via others#
[6]:
# read data
data = st.io.read_gef('./SS200000135TL_D1.tissue.gef')
# preprocessing
data.tl.filter_genes(gene_list=sct_high_genes.tolist())
data.tl.cal_qc()
data.tl.normalize_total()
data.tl.log1p()
data.tl.scale()
# embedding
data.tl.pca(use_highly_genes=False, hvg_res_key='highly_variable_genes', n_pcs=20, res_key='pca', svd_solver='arpack')
data.tl.neighbors(pca_res_key='pca', n_pcs=30, res_key='neighbors', n_jobs=8)
data.tl.umap(pca_res_key='pca', neighbors_res_key='neighbors', res_key='umap', init_pos='spectral', spread=2.0)
# clustering
data.tl.leiden(neighbors_res_key='neighbors', res_key='leiden')
completed 0 / 500 epochs
completed 50 / 500 epochs
completed 100 / 500 epochs
completed 150 / 500 epochs
completed 200 / 500 epochs
completed 250 / 500 epochs
completed 300 / 500 epochs
completed 350 / 500 epochs
completed 400 / 500 epochs
completed 450 / 500 epochs
Embedding comparison#
Display the UMAP distribution of them.
[7]:
data_sct.plt.umap(res_key='umap', cluster_key='leiden')
data.plt.umap(res_key='umap', cluster_key='leiden')
[7]:


Gene distribution#
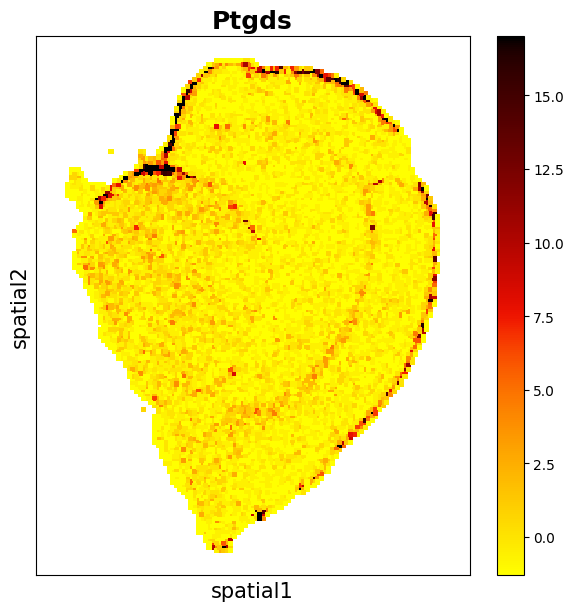
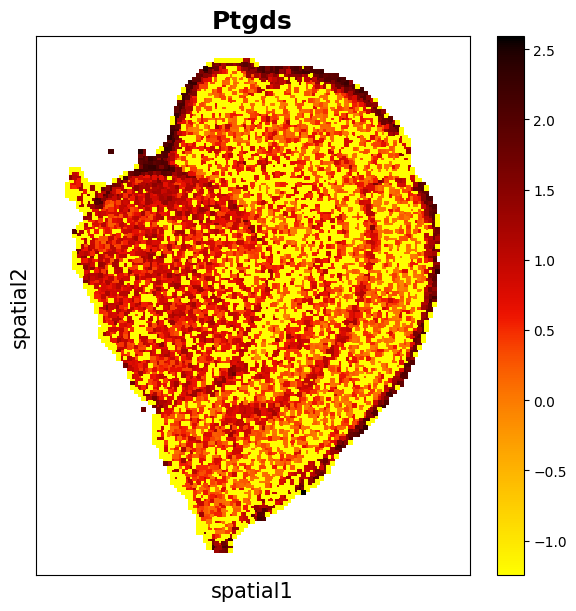
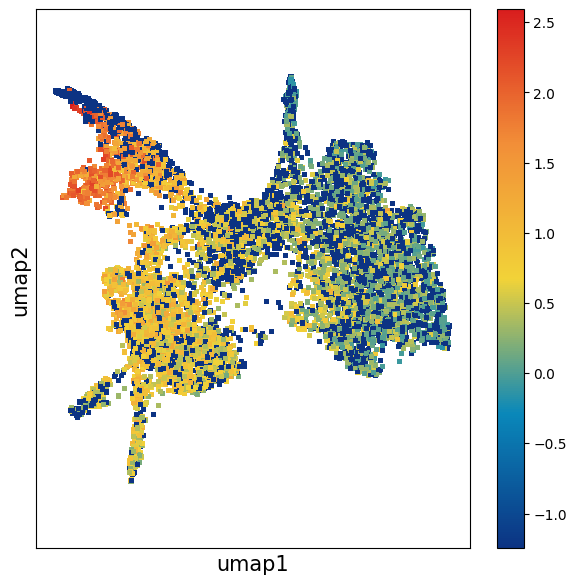
Observe the distribution of the gene Ptgds.
[8]:
data_sct.plt.umap(res_key='umap', gene_names=['Ptgds'])
data.plt.umap(res_key='umap', gene_names=['Ptgds'])
[8]:

[9]:
data_sct.plt.spatial_scatter_by_gene(gene_name='Ptgds')
data.plt.spatial_scatter_by_gene(gene_name='Ptgds')
[9]: